Anti-HBc total (Anti-Hepatitis B core total antibodies)
Useful For
Detection of antibodies (all classes) to Hepatitis B core antigen as a measure of previous infection.
Testing Algorithm
Reporting Name |
Available separately |
Always performed |
| Anti-HBc Total | NO | YES |
Indications for Testing
· Documenting prior exposure to Hepatitis B virus.
· Anti-HBc total should be ordered to document current/previous HBV infection
(see Special Instructions):
· Acute Hepatitis B follow-up
· Chronic Hepatitis Screen (unknown etiology)
· Previous Hepatitis Exposure Screen (unknown etiology)
Clinical Information
Anti-HBc total antibodies (all classes of antibodies) is the best marker for documenting prior exposure to HBV. Anti-HBc total antibodies become detectable before anti-HBs and remain positive indefinitely after IgM class anti-HBc disappears. In the absence of information about any other HBV markers, it must be considered that an individual with detectable levels of anti-HBc may be actively infected with HBV or that the infection may have resolved, leaving the person immune. Anti-HBc may be the only serological marker of HBV infection and potentially infectious blood. The presence of anti-HBc does not differentiate between acute or chronic hepatitis B infection. Anti-HBc should not be present in vaccinated individuals, unless they were infected with HBV prior to vaccination.
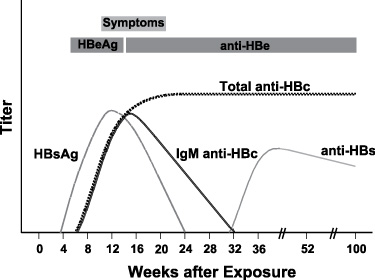
Typical sequence of serologic markers in patients with acute HBV infection with resolution of symptoms (Horvat, R. T., and Tegtmeier, G. E. 2007).
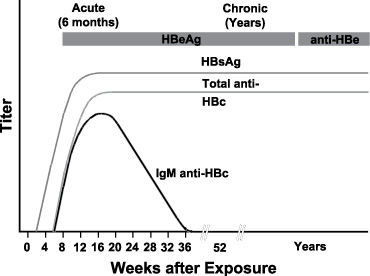
Typical sequence of serologic markers in patients with HBV infection that progresses to chronicity. In patients with chronic HBV infection, both HBsAg and IgG anti-HBc remain persistently detectable, generally for life. HBeAg is variably present in these patients (Horvat, R. T., and Tegtmeier, G. E. 2007).
Hepatitis B virus (HBV) causes a wide spectrum of manifestations ranging from asymptomatic seroconversion, sub-acute illness with non-specific symptoms (e.g. anorexia, nausea, or malaise) or extrahepatic symptoms, and clinical hepatitis with jaundice, to fuliminant fatal hepatitis. Only 10% of children and 50% of adults will exhibit symptoms. An acute illness may last up to three months with a fatality of 1-2%. In most acute cases HBsAg serum levels are positive initially, resolve and the individual develops anti-HBs which confers immunity.
HBV occurs worldwide, and is endemic in some Asian countries. In Canada the incidence of acute hepatitis B is estimated to be 2.3 per 100,000. In developed countries exposure to HBV may be common in certain high-risk groups such as injection drug users, person who have multiple sex partners, men who have sex with men, sex with HBV-infected persons and having a hepatitis B carrier in the family. The prevalence of chronic hepatitis B varies in different populations.
Chronic HBV infection is found in 0.5 % of adults in North America. After acute HBV infection, the risk of developing chronic infection varies with age; infants infected at birth have a 90% chance of becoming a chronic carrier. A chronic carrier is one who retains HBsAg positivity six months after the initial infection. These individuals are always infectious. Hepatocellular carcinoma and hepatic cirrhosis is likely to result in the premature death of 15-25% of those who have chronic HBV.
Reference Values
NON-REACTIVE
Interpretation
For diagnostic purposes, and to stage the disease, results should be used in conjunction with patient history and other hepatitis markers for diagnosis of acute or chronic infection (see special instructions).
REACTIVE: A REACTIVE result may indicate an acute or chronic infection. Anti-HBc antibodies remain detectable indefinitely after anti-HBc IgM disappears. In some patients anti-HBc may be the only serological marker for the detection of past infection in a patients with chronic inactive HBV.
NON-REACTIVE: A NON-REACTIVE result in the absence of other positive HBV markers indicates that the person has not been infected with HBV.
See Special Instructions for PHL recommended diagnostic approach to hepatitis.
Clinical Reference
Curry, M. P., and Chopra, S. 2010. Acute Viral Hepatitis, p. 1577-1592. In Mandell, D., Bennett, J. E., and Dolin, R. Principles and practice of infectious diseases, 7th ed., vol. 2. Churchill Livingstone, Elsevier, Philadelphia, PA.
Horvat, R. T., and Tegtmeier, G. E. 2007. Hepatitis B and D Viruses, p. 1641-1659. In Murray, P. R., Baron, E. J., Jorgensen, J. H., Landry, M. L., and Pfaller, M. A. Manual of Clinical Microbiology, 9th ed., vol. 2. ASM Press, American Society for Microbiology, Washington, DC.
James Koziel, M., and Thio, C. L. 2010. Hepatitis B Virus and Hepatitis Delta Virus, p. 2059-2086. In Mandell, D., Bennett, J. E., and Dolin, R. Principles and practice of infectious diseases, 7th ed., vol. 2. Churchill Livingstone, Elsevier, Philadelphia, PA.
Abbott. 2008. Architect System Anti-HBc II: package insert. Abbott, Diagnostics Division. Wiesbaden, Germany.